- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Bromhexine Hydrochloride |
 |
(Ph Eur monograph 0706)
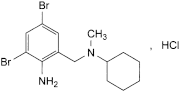
C14H20Br2N2,HCl 412.6 611-75-6
Mucolytic.
Ph Eur
N-(2-Amino-3,5-dibromobenzyl)-N-methylcyclohexanamine hydrochloride.
98.5 per cent to 101.5 per cent (dried substance).
White or almost white, crystalline powder.
Very slightly soluble in water, slightly soluble in alcohol and in methylene chloride.
It shows polymorphism (5.9).
First identification A, E.
Second identification B, C, D, E.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison bromhexine hydrochloride CRS.
If the spectra obtained in the solid state show differences, dissolve the substance to be examined and the reference substance separately in methanol R, evaporate to dryness and record new spectra using the residues.
B. Thin-layer chromatography (2.2.27).
Test solution Dissolve 20 mg of the substance to be examined in methanol R and dilute to 10 mL with the same solvent.
Reference solution Dissolve 20 mg of bromhexine hydrochloride CRS in methanol R and dilute to 10 mL with the same solvent.
Plate TLC silica gel F254 plate R.
Mobile phase glacial acetic acid R, water R, butanol R (17:17:66 V/V/V).
Application 20 µL.
Development Over 3/4 of the plate.
Drying In air.
Detection Examine in ultraviolet light at 254 nm.
Results The principal spot in the chromatogram obtained with the test solution is similar in position and size to the principal spot in the chromatogram obtained with the reference solution.
C. Dissolve about 25 mg in a mixture of 1 mL of dilute sulfuric acid R and 50 mL of water R. Add 2 mL of methylene chloride R and 5 mL of chloramine solution R and shake. A brownish-yellow colour develops in the lower layer.
D. Dissolve about 1 mg in 3 mL of 0.1 M hydrochloric acid. The solution gives the reaction of primary aromatic amines (2.3.1).
E. Dissolve about 20 mg in 1 mL of methanol R and add 1 mL of water R. The solution gives reaction (a) of chlorides (2.3.1).
The solution is clear (2.2.1) and not more intensely coloured than reference solution Y6 (2.2.2, Method II).
Dissolve 0.6 g in methanol R and dilute to 20 mL with the same solvent.
Liquid chromatography (2.2.29).
Test solution Dissolve 50 mg of the substance to be examined in methanol R and dilute to 10.0 mL with the same solvent.
Reference solution (a) Dissolve 5 mg of bromhexine impurity C CRS in methanol R, add 1.0 mL of the test solution and dilute to 10.0 mL with the same solvent.
Reference solution (b) Dilute 1.0 mL of the test solution to 100.0 mL with methanol R. Dilute 1.0 mL of this solution to 10.0 mL with methanol R.
- — size: l = 0.12 m, Ø = 4.6 mm,
- — stationary phase: end-capped octadecylsilyl silica gel for chromatography R (3 µm).
Mobile phase Mix 0.50 mL of phosphoric acid R in 950 mL of water R, adjust to pH 7.0 with triethylamine R (about 1.5 mL) and dilute to 1000 mL with water R; mix 20 volumes of this solution with 80 volumes of acetonitrile R.
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 248 nm.
Injection 10 µL.
Run time 2.5 times the retention time of bromhexine.
Relative retention With reference to bromhexine (retention time = about 11 min): impurity A = about 0.1; impurity B = about 0.2; impurity C = about 0.4; impurity D = about 0.5.
System suitability Reference solution (a):
- — resolution: minimum 12.0 between the peaks due to impurity C and bromhexine.
- — any impurity: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (b) (0.2 per cent), and not more than 1 such peak has an area greater than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.1 per cent),
- — total: not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent),
- — disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Maximum 1.0 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.300 g in 70 mL of alcohol R and add 1 mL of 0.1 M hydrochloric acid. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide. Read the volume between the 2 points of inflexion.
1 mL of 0.1 M sodium hydroxide is equivalent to 41.26 mg of C14H21Br2ClN2.
Protected from light.
Specified impurities A, B, C, D.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): E.
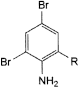
A. R = CH2OH: (2-amino-3,5-dibromophenyl)methanol,
B. R = CHO: 2-amino-3,5-dibromobenzaldehyde,
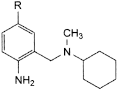
C. R = H: N-(2-aminobenzyl)-N-methylcyclohexanamine,
D. R = Br: N-(2-amino-5-bromobenzyl)-N-methylcyclohexanamine,
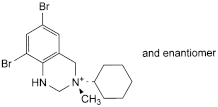
E. (3RS)-6,8-dibromo-3-cyclohexyl-3-methyl-1,2,3,4-tetrahydroquinazolin-3-ium.
Ph Eur

