- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Bronopol |
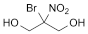
C3H6BrNO4 200.0 52-51-7
Antibacterial preservative.
Bronopol is 2-bromo-2-nitropropane-1,3-diol. It contains not less than 99.0% and not more than 101.0% of C3H6BrNO4, calculated with reference to the anhydrous substance.
White or almost white crystals or crystalline powder.
Freely soluble in water and in ethanol (96%); slightly soluble in glycerol and in liquid paraffin.
A. The infrared absorption spectrum, Appendix II A, is concordant with the reference spectrum of bronopol (RS 031).
B. Dissolve 0.1 g in 10 mL of water, add 10 mL of 7.5m sodium hydroxide and, carefully with constant stirring and cooling, 0.5 g of nickel-aluminium alloy. Allow the reaction to subside, filter and carefully neutralise with nitric acid. The resulting solution yields reaction A characteristic of bromides, Appendix VI.
C. Melting point, after drying over phosphorus pentoxide at a pressure not exceeding 0.7 kPa, about 130°, Appendix V A.
pH of a 1% w/v solution, 5.0 to 7.0, Appendix V L.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in the mobile phase.
(1) 0.2% w/v of the substance being examined.
(2) Dilute a volume of solution (1) to produce a solution containing 0.0002% w/v of the substance being examined.
(3) 0.001% w/v each of 2-methyl-2-nitropropan-1,3-diol and tris(hydroxymethyl)nitromethane.
(4) 0.0002% w/v each of 2-methyl-2-nitropropane-1,3-diol, 2-nitroethanol, sodium bromide and tris(hydroxymethyl)nitromethane and 0.2% w/v of the substance being examined.
(a) Use a stainless steel column (15 cm × 4.6 mm) packed with octadecylsilyl silica gel for chromatography (5 μm) (Phenomenex Luna C18 (2) is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 1 mL per minute.
(d) Use a column temperature of 35°.
(e) Use a detection wavelength of 214 nm.
(f) Inject 20 µL of each solution.
(g) For solution (1) allow the chromatography to proceed for at least 3 times the retention time of the principal peak.
1 volume of a 10% v/v solution of orthophosphoric acid, 10 volumes of acetonitrile and 189 volumes of water, adjust the pH to 3.0 using 2m sodium hydroxide.
The test is not valid unless, in the chromatogram obtained with solution (4):
the resolution factor between the peaks due to sodium bromide and tris(hydroxymethyl)nitromethane is at least 1.0;
the resolution factor between the peaks due to tris(hydroxymethyl)nitromethane and 2-nitroethanol is at least 1.5.
In the chromatogram obtained with solution (1):
the area of any peak corresponding to 2-methyl-2-nitropropane-1,3-diol and tris(hydroxymethyl)nitromethane are not greater than the area of the corresponding peaks in the chromatogram obtained with solution (3) (0.5% of each);
the area of any other secondary peak is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.1%).
Not more than 0.1%, Appendix IX A.
Not more than 0.5% w/w, Appendix IX C, Method I B. Use 5 g.
In a flask fitted with a reflux condenser dissolve 0.4 g in 15 mL of water and add 15 mL of 7.5m sodium hydroxide. Slowly, with caution, add 2 g of nickel-aluminium alloy through the reflux condenser, agitating the flask whilst cooling under running water. Allow the mixture to stand for 10 minutes and boil for 1 hour. Cool and filter under reduced pressure, washing the condenser, flask and residue with 150 mL of water. Combine the filtrate and washings, add 25 mL of nitric acid and 40 mL of 0.1m silver nitrate VS, shake vigorously and titrate with 0.1m ammonium thiocyanate VS using ammonium iron(iii) sulfate solution R2 as indicator. Repeat the operation without the substance being examined. The difference between the titrations represents the amount of silver nitrate required. Each mL of 0.1m silver nitrate VS is equivalent to 20.00 mg of C3H6BrNO4.
Bronopol should be protected from light.

