- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Clomethiazole |
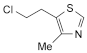
C6H8ClNS 161.6 533-45-9
Hypnotic.
Clomethiazole is 5-(2-chloroethyl)-4-methyl-thiazole. It contains not less than 98.0% and not more than 101.0% of C6H8ClNS.
A colourless to slightly yellowish brown liquid.
Slightly soluble in water; miscible with ethanol (96%) and with ether.
A. The light absorption, Appendix II B, in the range 230 to 350 nm of a 0.004% w/v solution in 0.1m hydrochloric acid exhibits a maximum only at 257 nm. The absorbance at the maximum is about 1.1.
B. The infrared absorption spectrum, Appendix II A, is concordant with the reference spectrum of clomethiazole (RS 051).
C. Mix 0.1 g with 0.2 g of powdered sodium hydroxide, heat to fusion and continue heating for a further few seconds. Cool, add 0.5 mL of water and a slight excess of 2m hydrochloric acid and warm. Any fumes evolved do not turn moistened starch iodate paper blue (distinction from clomethiazole edisilate).
pH of a 0.5% w/v solution, 5.5 to 7.0, Appendix V L.
Moisten the residue obtained in the test for Sulfated ash with 2 mL of hydrochloric acid and evaporate to dryness. Dissolve the residue in water and add sufficient water to produce 20 mL. 12 mL of the resulting solution complies with limit test A for heavy metals, Appendix VII. Use lead standard solution (1 ppm Pb) to prepare the standard (20 ppm).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. Solution (1) contains 0.20% w/v of the substance being examined in the mobile phase. For solution (2) dilute 1 volume of solution (1) to 1000 volumes with the mobile phase. For solution (3) dilute 1 volume of a 0.030% w/v solution of 4-methyl-5-vinylthiazole edisilate BPCRS in methanol (solution A) to 50 volumes with the mobile phase. For solution (4) dilute 1 volume of a 0.020% w/v solution of 5-(2-chloroethyl)-4-methyl-3-[2-(4-methylthiazol-5-yl)ethyl]-thiazolium chloride BPCRS (quaternary dimer) in methanol (solution B) to 50 volumes with the mobile phase. For solution (5) dilute 1 volume of a 0.020% w/v solution of 4-methyl-5-(2-hydroxyethyl)thiazole BPCRS in methanol (solution C) to 50 volumes with the mobile phase. For solution (6) add 1 mL each of solutions A, B and C to 0.10 g of the substance being examined and dilute to 50 mL with the mobile phase.
The chromatographic procedure may be carried out using (a) a stainless steel column (20 cm × 4 mm) packed with octadecylsilyl silica gel for chromatography (10 µm) (Lichrosorb RP18 is suitable), (b) as the mobile phase with a flow rate of 1 mL per minute, a mixture of 70 volumes of a solution containing 0.13% w/v of sodium hexanesulfonate and 2.7% w/v of tetramethylammonium hydrogen sulfate, adjusted to pH 2.0 with 5m sodium hydroxide, and 30 volumes of methanol and (c) a detection wavelength of 257 nm.
The test is not valid unless in the chromatogram obtained with solution (6) baseline separation is achieved between the peaks due to the three specified impurities and also between the principal peak and the two adjacent specified impurity peaks.
Calculate the content of each of the three specified impurities in the substance being examined expressing the content of 4-methyl-5-vinylthiazole as the base (1 mg of 4-methyl-5-vinylthiazole edisilate is equivalent to 0.568 mg of base). The total content of the three specified impurities is not greater than 0.5%. In the chromatogram obtained with solution (1) the area of any other secondary peak is not greater than the area of the peak in the chromatogram obtained with solution (2).
Not more than 0.1%, Appendix IX A. Use 1 g.
Carry out Method I for non-aqueous titration, Appendix VIII A, using 0.3 g and determining the end point potentiometrically. Each mL of 0.1m perchloric acid VS is equivalent to 16.16 mg of C6H8ClNS.
Clomethiazole should be stored at a temperature of 2° to 8°.

