- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Cyproterone Acetate |
 |
(Ph Eur monograph 1094)
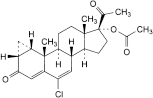
C24H29ClO4 416.9 427-51-0
Anti-estrogen.
Ph Eur
6-Chloro-3,20-dioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4,6-trien-17-yl acetate.
97.0 per cent to 103.0 per cent (dried substance).
White or almost white, crystalline powder.
Practically insoluble in water, very soluble in methylene chloride, freely soluble in acetone, soluble in methanol, sparingly soluble in anhydrous ethanol.
About 210 °C.
First identification A.
Second identification B, C, D, E.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison cyproterone acetate CRS.
B. Thin-layer chromatography (2.2.27).
Test solution Dissolve 20 mg of the substance to be examined in methylene chloride R and dilute to 10 mL with the same solvent.
Reference solution Dissolve 10 mg of cyproterone acetate CRS in methylene chloride R and dilute to 5 mL with the same solvent.
Plate TLC silica gel F254 plate R.
Mobile phase cyclohexane R, ethyl acetate R (50:50 V/V).
Application 5 µL.
Development Twice over 3/4 of the plate; dry in air between the 2 developments.
Drying In air.
Detection Examine in ultraviolet light at 254 nm.
Results The principal spot in the chromatogram obtained with the test solution is similar in position and size to the principal spot in the chromatogram obtained with the reference solution.
C. To about 1 mg add 2 mL of sulfuric acid R and heat on a water-bath for 2 min. A red colour develops. Cool. Add this solution cautiously to 4 mL of water R and shake. The solution becomes violet.
D. Incinerate about 30 mg with 0.3 g of anhydrous sodium carbonate R over a naked flame for about 10 min. Cool and dissolve the residue in 5 mL of dilute nitric acid R. Filter. To 1 mL of the filtrate add 1 mL of water R. The solution gives reaction (a) of chlorides (2.3.1).
E. It gives the reaction of acetyl (2.3.1).
+ 152 to + 157 (dried substance).
Dissolve 0.25 g in acetone R and dilute to 25.0 mL with the same solvent.
Liquid chromatography (2.2.29).
Test solution Dissolve 10 mg of the substance to be examined in acetonitrile R and dilute to 10.0 mL with the same solvent.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with acetonitrile R.
Reference solution (b) Dissolve the contents of a vial of cyproterone impurity mixture CRS (impurities F and I) in 1.0 mL of the test solution.
Reference solution (c) Dissolve 2 mg of cyproterone acetate for peak identification CRS (containing impurities B, C, E and G) in 2.0 mL of acetonitrile R.
- — size: l = 0.125 m, Ø = 4.6 mm;
- — stationary phase: end-capped octadecylsilyl silica gel for chromatography R (3 µm).
Mobile phase acetonitrile R, water R (40:60 V/V).
Flow rate 1.5 mL/min.
Detection Spectrophotometer at 254 nm.
Injection 20 µL.
Run time Twice the retention time of cyproterone acetate.
Identification of impurities Use the chromatogram supplied with cyproterone impurity mixture CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities F and I; use the chromatogram supplied with cyproterone acetate for peak identification CRS and the chromatogram obtained with reference solution (c) to identify the peaks due to impurities B, C, E and G.
Relative retention With reference to cyproterone acetate (retention time = about 22 min): impurity E = about 0.27; impurity G = about 0.3; impurity F = about 0.5; impurity B = about 0.7; impurity I = about 0.9; impurity C = about 1.5.
System suitability Reference solution (b):
- — resolution: minimum 1.5 between the peaks due to impurity I and cyproterone acetate.
- — correction factors: for the calculation of content, multiply the peak areas of the following impurities by the corresponding correction factor: impurity C = 1.8; impurity E = 0.7;
- — impurity F: not more than 0.4 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.4 per cent);
- — impurity E: not more than 0.2 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
- — impurities B, C, G: for each impurity, not more than 0.15 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.15 per cent);
- — unspecified impurities: for each impurity, not more than 0.1 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
- — total: not more than 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent);
- — disregard limit: 0.05 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Maximum 0.5 per cent, determined on 1.000 g by drying at 80 °C at a pressure not exceeding 0.7 kPa.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 50.0 mg in methanol R and dilute to 50.0 mL with the same solvent. Dilute 1.0 mL of the solution to 100.0 mL with methanol R. Measure the absorbance (2.2.25) at the absorption maximum at 282 nm.
Calculate the content of C24H29ClO4 taking the specific absorbance to be 414.
Protected from light.
Specified impurities B, C, E, F, G.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): A, D, H, I, J.
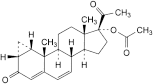
A. 3,20-dioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4,6-trien-17-yl acetate,
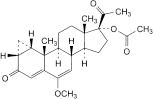
B. 6-methoxy-3,20-dioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4,6-trien-17-yl acetate,
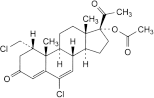
C. 6-chloro-1α-(chloromethyl)-3,20-dioxopregna-4,6-dien-17-yl acetate,
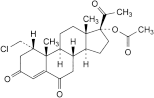
D. 1α-(chloromethyl)-3,6,20-trioxopregn-4-en-17-yl acetate,
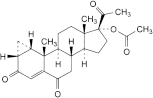
E. 3,6,20-trioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4-dien-17-yl acetate,
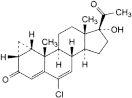
F. 6-chloro-17-hydroxy-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4,6-triene-3,20-dione,
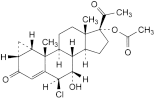
G. 6β-chloro-7α-hydroxy-3,20-dioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4-dien-17-yl acetate,
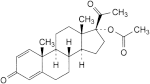
H. 3,20-dioxopregna-1,4-dien-17-yl acetate,
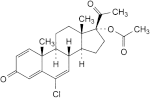
I. 6-chloro-3,20-dioxopregna-1,4,6-trien-17-yl acetate (delmadinone acetate),
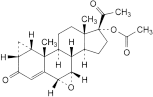
J. 6α,7α-epoxy-3,20-dioxo-1β,2β-dihydro-3′H-cyclopropa[1,2]pregna-1,4-dien-17-yl acetate.
Ph Eur

