- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Ethanol (96 per cent) |
 |
Alcohol (96 per cent)
(Ph. Eur. monograph 1317)
Ph Eur
- — ethanol (C2H6O; Mr 46.07): 95.1 per cent V/V (92.6 per cent m/m) to 96.9 per cent V/V (95.2 per cent m/m) at 20 °C, calculated from the relative density using the alcoholimetric tables (5.5);
- — water.
Colourless, clear, volatile, flammable liquid, hygroscopic.
Miscible with water and with methylene chloride.
It burns with a blue, smokeless flame.
About 78 °C.
First identification A, B.
Second identification A, C, D.
A. Relative density (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison Ph. Eur. reference spectrum ethanol (96 per cent).
C. Mix 0.1 mL with 1 mL of a 10 g/L solution of potassium permanganate R and 0.2 mL of dilute sulfuric acid R in a test-tube. Cover immediately with a filter paper moistened with a freshly prepared solution containing 0.1 g of sodium nitroprusside R and 0.5 g of piperazine hydrate R in 5 mL of water R. After a few minutes, an intense blue colour appears on the paper and becomes paler after 10-15 min.
D. To 0.5 mL add 5 mL of water R, 2 mL of dilute sodium hydroxide solution R, then slowly add 2 mL of 0.05 M iodine. A yellow precipitate is formed within 30 min.
It is clear (2.2.1) and colourless (2.2.2, Method II) when compared with water R. Dilute 1.0 mL to 20 mL with water R. After standing for 5 min, the dilution remains clear (2.2.1) when compared with water R.
To 20 mL add 20 mL of carbon dioxide-free water R and 0.1 mL of phenolphthalein solution R. The solution is colourless. Add 1.0 mL of 0.01 M sodium hydroxide. The solution is pink (30 ppm, expressed as acetic acid).
0.805 to 0.812.
Maximum 0.40 at 240 nm, 0.30 between 250 nm and 260 nm and 0.10 between 270 nm and 340 nm. The absorption curve is smooth.
Examine between 235 nm and 340 nm, in a 5 cm cell using water R as the compensation liquid.
Gas chromatography (2.2.28).
Test solution (a) The substance to be examined.
Test solution (b) Add 150 µL of 4-methylpentan-2-ol R to 500.0 mL of the substance to be examined.
Reference solution (a) Dilute 100 µL of anhydrous methanol R to 50.0 mL with the substance to be examined. Dilute 5.0 mL of the solution to 50.0 mL with the substance to be examined.
Reference solution (b) Dilute 50 µL of anhydrous methanol R and 50 µL of acetaldehyde R to 50.0 mL with the substance to be examined. Dilute 100 µL of the solution to 10.0 mL with the substance to be examined.
Reference solution (c) Dilute 150 µL of acetal R to 50.0 mL with the substance to be examined. Dilute 100 µL of the solution to 10.0 mL with the substance to be examined.
Reference solution (d) Dilute 100 µL of benzene R to 100.0 mL with the substance to be examined. Dilute 100 µL of the solution to 50.0 mL with the substance to be examined.
- — material: fused silica;
- — size: l = 30 m, Ø = 0.32 mm;
- — stationary phase: poly[(cyanopropyl)(phenyl)][dimethyl]siloxane R (film thickness 1.8 µm).
Carrier gas helium for chromatography R.
Linear velocity 35 cm/s.
Split ratio 1:20.
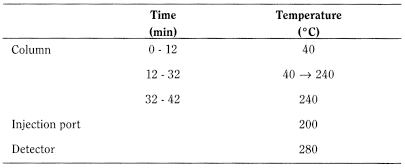
Detection Flame ionisation.
Injection 1 µL.
System suitability Reference solution (b):
- — resolution: minimum 1.5 between the first peak (acetaldehyde) and the second peak (methanol).
- — methanol in the chromatogram obtained with test solution (a): not more than half the area of the corresponding peak in the chromatogram obtained with reference solution (a) (200 ppm V/V);
- — acetaldehyde + acetal: maximum 10 ppm V/V, expressed as acetaldehyde.
Calculate the sum of the contents of acetaldehyde and acetal in parts per million (V/V) using the following expression:
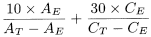
A E |
= |
area of the acetaldehyde peak in the chromatogram obtained with test solution (a), |
A T |
= |
area of the acetaldehyde peak in the chromatogram obtained with reference solution (b), |
C E |
= |
area of the acetal peak in the chromatogram obtained with test solution (a), |
C T |
= |
area of the acetal peak in the chromatogram obtained with reference solution (c). |
- — benzene: maximum 2 ppm V/V.
Calculate the content of benzene in parts per million (V/V) using the following expression:
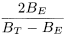
B E |
= |
area of the benzene peak in the chromatogram obtained with the test solution (a), |
B T |
= |
area of the benzene peak in the chromatogram obtained with reference solution (d). |
If necessary, the identity of benzene can be confirmed using another suitable chromatographic system (stationary phase with a different polarity).
- — total of other impurities in the chromatogram obtained with test solution (b): not more than the area of the peak due to 4-methylpentan-2-ol in the chromatogram obtained with test solution (b) (300 ppm),
- — disregard limit: 0.03 times the area of the peak corresponding to 4-methylpentan-2-ol in the chromatogram obtained with test solution (b) (9 ppm).
Maximum 25 ppm m/V.
Evaporate 100 mL to dryness on a water-bath and dry at 100-105 °C for 1 h. The residue weighs a maximum of 2.5 mg.
Protected from light.
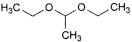
A. 1,1-diethoxyethane (acetal),
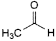
B. acetaldehyde,
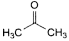
C. propan-2-one (acetone),
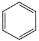
D. benzene,
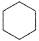
E. cyclohexane,
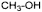
F. methanol,
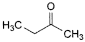
G. butan-2-one (methyl ethyl ketone),
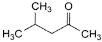
H. 4-methylpentan-2-one (methyl isobutyl ketone),

I. propan-1-ol (propanol),
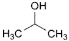
J. propan-2-ol (isopropyl alcohol),

K. butan-1-ol (butanol),
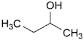
L. butan-2-ol,
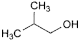
M. 2-methylpropan-1-ol (isobutanol),
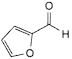
N. furane-2-carbaldehyde (furfural),
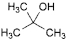
O. 2-methylpropan-2-ol (1,1-dimethylethyl alcohol),
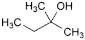
P. 2-methylbutan-2-ol,
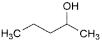
Q. pentan-2-ol,

R. pentan-1-ol (pentanol),

S. hexan-1-ol (hexanol),
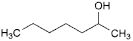
T. heptan-2-ol,
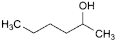
U. hexan-2-ol,
V. hexan-3-ol.
Ph Eur

