- British Pharmacopoeia Volume III
- Formulated Preparations: Specific Monographs
Tranexamic Acid Injection |
Antifibrinolytic.
Tranexamic Acid Injection is a sterile solution of Tranexamic Acid in Water for Injections.
The injection complies with the requirements stated under Parenteral Preparations and with the following requirements.
95.0 to 105.0% of the stated amount.
To a volume containing 0.4 g of Tranexamic Acid add 2 mL of ether, stir, add 5 mL of methanol, stir again and allow to crystallise. The crystals, after drying, comply with the following tests.
A. The infrared absorption spectrum, Appendix II A, is concordant with the reference spectrum of tranexamic acid (RS 344).
B. To 1 mL of a 1% w/v solution add 1 mL of a 0.2% w/v solution of ninhydrin in ethanol (96%) and heat on a water bath for 2 minutes. A dark bluish violet colour is produced.
C. Dissolve 0.2 g in 10 mL of 5m sodium hydroxide, add 0.2 mL of benzoyl chloride and shake vigorously for 10 minutes. Acidify to pH 4 with 2m hydrochloric acid, filter, wash the residue with 5 mL of ether and dry at 50° at a pressure of 2 kPa. The melting point of the residue is about 186°, Appendix V A.
pH, 6.5 to 8.0, Appendix V L.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
(1) Dilute the injection, if necessary, with sufficient water to produce a solution containing 1% w/v of Tranexamic Acid.
(2) 0.01% w/v of tranexamic acid EPCRS in water.
(3) 0.001% w/v of 4-aminomethylbenzoic acid in water.
(4) 1% w/v of tranexamic acid impurity standard BPCRS in water.
(a) Use a stainless steel column (25 cm × 4.6 mm) packed with octadecylsilyl silica gel for chromatography (5 µm) (Hypersil ODS is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 0.9 mL per minute.
(d) Use an ambient column temperature.
(e) Use a detection wavelength of 220 nm.
(f) Inject 20 µL of each solution.
Inject solution (1) and allow the chromatography to proceed for 3 times the retention time of tranexamic acid (about 13 minutes). Identify the impurities from the chromatogram obtained with solution (4) and from the reference chromatogram supplied with tranexamic acid impurity standard BPCRS.
Dissolve 11.0 g of anhydrous sodium dihydrogen orthophosphate in 500 mL of water, add 5 mL of triethylamine and 1.4 g of sodium dodecyl sulfate, adjust the pH to 2.5 with 2m orthophosphoric acid and add sufficient water to produce 600 mL. Add 400 mL of methanol and mix.
The test is not valid unless:
the chromatogram obtained with solution (4) closely resembles the reference chromatogram supplied with tranexamic acid impurity standard BPCRS;
in the chromatogram obtained with solution (4) the resolution factor between the peaks corresponding to tranexamic acid and impurity C is at least 2.0.
In the chromatogram obtained with solution (1):
the area of any peak corresponding to impurity A is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1%);
the area of any peak corresponding to impurity B is not greater than 0.5 times the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
the area of any peak corresponding to impurity C is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.1%);
the area of any peak corresponding to impurity D is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.1%);
the area of any other secondary peak is not greater than 0.1 times the area of the principal peak in the chromatogram obtained with solution (2) (0.1%).
Carry out the test for bacterial endotoxins, Appendix XIV C. The endotoxin limit concentration is less than 35 IU per mL of Tranexamic Acid Injection.
To a volume containing 0.1 g of Tranexamic Acid add 50 mL of water and adjust the solution to pH 7.0 with 0.1m sodium hydroxide or 0.1m hydrochloric acid. Add 25 mL of formaldehyde solution, previously adjusted to pH 7.0, and 20 mL of 0.1m sodium hydroxide VS and titrate with 0.1m hydrochloric acid VS determining the end point potentiometrically. Repeat the operation without the injection. The difference between the titrations represents the amount of sodium hydroxide required. Each mL of 0.1m sodium hydroxide VS is equivalent to 15.72 mg of C8H15NO2.
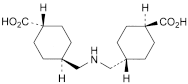
A. trans,trans-4,4′-(iminodimethylene)di(cyclohexanecarboxylic) acid,
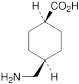
B. cis-4-(aminomethyl)cyclohexanecarboxylic acid,
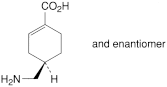
C. (RS)-4-(aminomethyl)cyclohex-1-enecarboxylic acid,
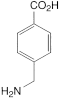
D. 4-aminomethylbenzoic acid.

