- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Cefadroxil Monohydrate |
 |
(Ph. Eur. monograph 0813)
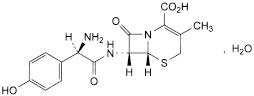
C16H17N3O5S,H2O 381.4 66592-87-8
Cephalosporin antibacterial.
Ph Eur
(6R,7R)-7-[[(2R)-2-Amino-2-(4-hydroxyphenyl)acetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid monohydrate.
Semi-synthetic product derived from a fermentation product.
95.0 per cent to 102.0 per cent (anhydrous substance).
White or almost white powder.
Slightly soluble in water, very slightly soluble in ethanol (96 per cent).
Infrared absorption spectrophotometry (2.2.24).
Comparison cefadroxil CRS.
4.0 to 6.0.
Suspend 1.0 g in carbon dioxide-free water R and dilute to 20 mL with the same solvent.
+ 165 to + 178 (anhydrous substance).
Dissolve 0.500 g in water R and dilute to 50.0 mL with the same solvent.
Liquid chromatography (2.2.29).
Test solution Dissolve 50.0 mg of the substance to be examined in mobile phase A and dilute to 50.0 mL with mobile phase A.
Reference solution (a) Dissolve 10.0 mg of d-α-(4-hydroxyphenyl)glycine CRS (impurity A) in mobile phase A and dilute to 10.0 mL with mobile phase A.
Reference solution (b) Dissolve 10.0 mg of 7-aminodesacetoxycephalosporanic acid CRS (impurity B) in phosphatebuffer solution pH 7.0 R5 and dilute to 10.0 mL with the same buffer solution.
Reference solution (c) Dilute 1.0 mL of reference solution (a) and 1.0 mL of reference solution (b) to 100.0 mL with mobile phase A.
Reference solution (d) Dissolve 10 mg of dimethylformamide R and 10 mg of dimethylacetamide R in mobile phase A and dilute to 10.0 mL with mobile phase A. Dilute 1.0 mL of this solution to 100.0 mL with mobile phase A.
Reference solution (e) Dilute 1.0 mL of reference solution (c) to 25.0 mL with mobile phase A.
- — size: l = 0.10 m, Ø = 4.6 mm,
- — stationary phase: spherical octadecylsilyl silica gel for chromatography R (5 µm).
- — mobile phase A: phosphate buffer solution pH 5.0 R,
- — mobile phase B: methanol R2,

Flow rate 1.5 mL/min.
Detection Spectrophotometer at 220 nm.
Injection 20 µL of the test solution and reference solutions (c), (d) and (e).
Relative retention With reference to cefadroxil (retention time = about 6 min): dimethylformamide = about 0.4; dimethylacetamide = about 0.75.
- — resolution: minimum 5.0 between the peaks due to impurities A and B in the chromatogram obtained with reference solution (c),
- — signal-to-noise ratio: minimum 10 for the 2nd peak in the chromatogram obtained with reference solution (e).
- — impurity A: not more than the area of the 1st peak in the chromatogram obtained with reference solution (c) (1.0 per cent),
- — any other impurity: for each impurity, not more than the area of the 2nd peak in the chromatogram obtained with reference solution (c) (1.0 per cent),
- — total: not more than 3 times the area of the 2nd peak in the chromatogram obtained with reference solution (c) (3.0 per cent),
- — disregard limit: 0.05 times the area of the 2nd peak in the chromatogram obtained with reference solution (c) (0.05 per cent); disregard the peaks due to dimethylformamide and dimethylacetamide.
Maximum 20 ppm.
4.0 per cent to 6.0 per cent, determined on 0.200 g.
Maximum 0.5 per cent, determined on 1.0 g.
Liquid chromatography (2.2.29).
Test solution Dissolve 50.0 mg of the substance to be examined in the mobile phase and dilute to 100.0 mL with the mobile phase.
Reference solution (a) Dissolve 50.0 mg of cefadroxil CRS in the mobile phase and dilute to 100.0 mL with the mobile phase.
Reference solution (b) Dissolve 5 mg of cefadroxil CRS and 50 mg of amoxicillin trihydrate CRS in the mobile phase and dilute to 100 mL with the mobile phase.
- — size: l = 0.25 m, Ø = 4.6 mm,
- — stationary phase: octadecylsilyl silica gel for chromatography R (5 µm).
Mobile phase acetonitrile R, a 2.72 g/L solution of potassium dihydrogen phosphate R (4:96 V/V).
Flow rate 1 mL/min.
Detection Spectrophotometer at 254 nm.
Injection 20 µL.
System suitability Reference solution (b):
- — resolution: minimum 5.0 between the peaks due to cefadroxil and to amoxicillin.
Calculate the percentage content of cefadroxil.
Protected from light.
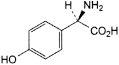
A. (2R)-2-amino-2-(4-hydroxyphenyl)acetic acid,
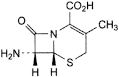
B. (6R,7R)-7-amino-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-ADCA),
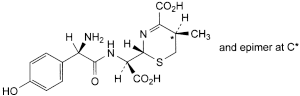
C. (2R,5RS)-2-[(R)-[[(2R)-2-amino-2-(4-hydroxyphenyl)acetyl]amino]carboxymethyl]-5-methyl-5,6-dihydro-2H-1,3-thiazine-4-carboxylic acid,
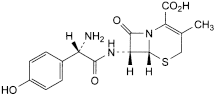
D. (6R,7R)-7-[[(2S)-2-amino-2-(4-hydroxyphenyl)acetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (l-cefadroxil),
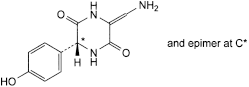
E. (6RS)-3-(aminomethylene)-6-(4-hydroxyphenyl)piperazine-2,5-dione,
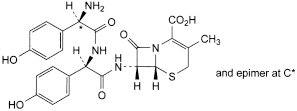
F. (6R,7R)-7-[[(2R)-2-[[(2RS)-2-amino-2-(4-hydroxyphenyl)acetyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,
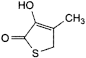
G. 3-hydroxy-4-methylthiophen-2(5H)-one,
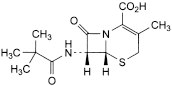
H. (6R,7R)-7-[(2,2-dimethylpropanoyl)amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-ADCA pivalamide).
Ph Eur