- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Mefenamic Acid |
 |
(Ph. Eur. monograph 1240)
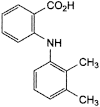
C15H15NO2 241.3 61-68-7
Cyclo-oxygenase inhibitor; analgesic; anti-inflammatory.
Ph Eur
2-[(2,3-Dimethylphenyl)amino]benzoic acid.
99.0 per cent to 101.0 per cent (dried substance).
White or almost white, microcrystalline powder.
Practically insoluble in water, slightly soluble in ethanol (96 per cent) and in methylene chloride. It dissolves in dilute solutions of alkali hydroxides.
It shows polymorphism (5.9).
Infrared absorption spectrophotometry (2.2.24).
Comparison mefenamic acid CRS.
If the spectra obtained in the solid state show differences, dissolve the substance to be examined and the reference substance separately in ethanol (96 per cent) R, evaporate to dryness and record new spectra using the residues.
Liquid chromatography (2.2.29).
Test solution Dissolve 25.0 mg of the substance to be examined in the mobile phase and dilute to 25.0 mL with the mobile phase.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (b) Dissolve 50 mg of 2-chlorobenzoic acid R (impurity C) and 50 mg of benzoic acid R (impurity D) in the mobile phase and dilute to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 100.0 mL with the mobile phase.
Reference solution (c) Dissolve 10.0 mg of mefenamic acid impurity A CRS in the mobile phase and dilute to 10.0 mL with the mobile phase. Dilute 1.0 mL of the solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 100.0 mL with the mobile phase.
Reference solution (d) Dissolve 20.0 mg of benzoic acid R in the mobile phase and dilute to 1000.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 100.0 mL with the mobile phase.
- — size: l = 0.25 m, Ø = 4.6 mm;
- — stationary phase: spherical octadecylsilyl silica gel for chromatography R (5 µm).
Mobile phase Mix 14 volumes of tetrahydrofuran R, 40 volumes of a 5.75 g/L solution of ammonium dihydrogen phosphate R adjusted to pH 5.0 with dilute ammonia R2, and 46 volumes of acetonitrile R1.
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 254 nm.
Injection 10 µL.
Run time 4 times the retention time of mefenamic acid.
Identification of impurities Use the chromatogram obtained with reference solution (b) to identify the peaks due to impurities C and D.
Relative retention With reference to mefenamic acid (retention time = about 8 min): impurity C = about 0.3; impurity D = about 0.35; impurity A = about 0.5.
- — resolution: minimum 3.0 between the peaks due to impurities C and D in the chromatogram obtained with reference solution (b);
- — signal-to-noise ratio: minimum 10 for the principal peak in the chromatogram obtained with reference solution (d).
- — correction factors: for the calculation of content, multiply the peak areas of the following impurities by the corresponding correction factor: impurity C = 5.9; impurity D = 4.0;
- — impurities C, D: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent);
- — impurity A: not more than the area of the corresponding peak in the chromatogram obtained with reference solution (c) (100 ppm);
- — unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
- — total: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
- — disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent); disregard the peak due to impurity A.
Maximum 10 ppm.
Atomic absorption spectrometry (2.2.23, Method I).
Test solution Place 1.00 g in a silica crucible, moisten with sulfuric acid R, heat cautiously on a flame for 30 min and then progressively to 650 °C. Continue ignition until all black particles have disappeared. Allow to cool, dissolve the residue in 0.1 M hydrochloric acid and dilute to 25.0 mL with the same acid.
Reference solutions Prepare the reference solutions using copper standard solution (0.1 per cent Cu) R, diluting with 0.1 M nitric acid.
Source Copper hollow-cathode lamp.
Wavelength 324.8 nm.
Atomisation device Air-acetylene flame.
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve with the aid of ultrasound 0.200 g in 100 mL of warm anhydrous ethanol R, previously neutralised to phenol red solution R. Add 0.1 mL of phenol red solution R and titrate with 0.1 M sodium hydroxide.
1 mL of 0.1 M sodium hydroxide is equivalent to 24.13 mg of C15H15NO2.
Specified impurities A, C, D.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): B, E.
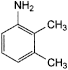
A. 2,3-dimethylaniline,
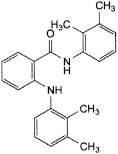
B. N-(2,3-dimethylphenyl)-2-[(2,3-dimethylphenyl)amino]benzamide,
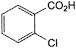
C. 2-chlorobenzoic acid,
D. benzoic acid,
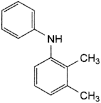
E. 2,3-dimethyl-N-phenylaniline.
Ph Eur