- British Pharmacopoeia Volume III
- Formulated Preparations: General Monographs
Tablets of the British Pharmacopoeia |
In addition to the above requirements of the European Pharmacopoeia, the following statements apply to those tablets that are the subject of an individual monograph in the British Pharmacopoeia.
Tablets of the British Pharmacopoeia may contain flavouring only when indicated in the individual monograph.
Tablets that are the subject of individual monographs in the British Pharmacopoeia may be uncoated, compression-coated, film-coated or sugar-coated unless otherwise indicated in the monograph. Tablets for which the monograph states 'They are coated' are compression-coated or film-coated or sugar-coated. Tablets may be made gastro-resistant by enteric-coating or by other means only where this is specifically indicated in the monograph.
When presented as coated formulations, where justified and authorised, it may be necessary to remove the coating before performing a test described in a monograph. Removal of a coating is not permitted where it affects the functionality of the product, for example in dissolution or disintegration tests.
In preparing Tablets for which Chocolate Basis is specified, the active ingredient may be incorporated with a mixture of 15 parts of non-alkalinised cocoa powder of commerce, 15 parts of Sucrose and 70 parts of Lactose.
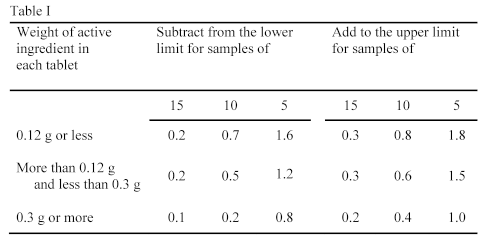
The range for the content of active ingredient stated in the monograph is based on the requirement that 20 tablets, or such other number as may be indicated in the monograph, are used in the Assay. In circumstances where 20 tablets cannot be obtained, a smaller number, which must not be less than five, may be used, but to allow for sampling errors the tolerances are widened in accordance with Table I.
The requirements of Table I apply when the stated limits are 90 to 110%. For limits other than 90 to 110%, proportionately smaller or larger allowances should be made.
Comply with the disintegration test for tablets and capsules, Appendix XII A1, unless otherwise stated in the individual monographs.
For those Uncoated or Coated Tablets for which a requirement for Dissolution is included in the individual monograph, the requirement for Disintegration does not apply.
Details of the analytical method to be employed for determining the content of active ingredient may be included in certain monographs. Unless otherwise stated in the monograph the limits are as given in test A for Uniformity of content, Appendix XII C3.
Any tablets that, when examined individually, show a gross deviation from the stated content are not official.