- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Temazepam |
 |
(Ph Eur monograph 0954)
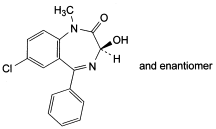
C16H13ClN2O2 300.7 846-50-4
Benzodiazepine.
Ph Eur
(3RS)-7-Chloro-3-hydroxy-1-methyl-5-phenyl-1,3-dihydro-2H-
1,4-benzodiazepin-2-one.
99.0 per cent to 101.0 per cent (dried substance).
White or almost white, crystalline powder.
Practically insoluble in water, freely soluble in methylene chloride, sparingly soluble in ethanol (96 per cent).
Infrared absorption spectrophotometry (2.2.24).
Comparison temazepam CRS.
Maximum 0.05 per cent.
Dissolve 0.400 g in methylene chloride R and dilute to 20.0 ml with the same solvent. The absorbance (2.2.25) is not greater than 0.30 at 409 nm.
Liquid chromatography (2.2.29).
Test solution Dissolve 10.0 mg of the substance to be examined in a mixture of 1 volume of water R and 9 volumes of methanol R and dilute to 50.0 ml with the same mixture of solvents.
Reference solution (a) Dilute 1.0 ml of the test solution to 100.0 ml with a mixture of 1 volume of water R and 9 volumes of methanol R. Dilute 2.0 ml of this solution to 10.0 ml with a mixture of 1 volume of water R and 9 volumes of methanol R.
Reference solution (b) Dissolve 1 mg of oxazepam R, 1 mg of temazepam impurity F CRS and 1 mg of temazepam impurity G CRS in a mixture of 1 volume of water R and 9 volumes of methanol R and dilute to 25 ml with the same mixture of solvents.
Reference solution (c) Dissolve 1 mg of temazepam impurity C CRS and 1 mg of temazepam impurity D CRS with a mixture of 1 volume of water R and 9 volumes of methanol R and dilute to 25 ml with the same mixture of solvents.
- — size: l = 0.15 m, Ø = 4.6 mm;
- — stationary phase: end-capped octadecylsilyl silica gel for chromatography R (3.5 µm).
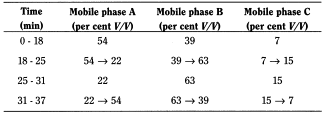
- — mobile phase A: solution containing 4.9 g/l of sodium dihydrogen phosphate R and 0.63 g/l of disodium hydrogen phosphate R (pH 5.6);
- — mobile phase B: methanol R;
- — mobile phase C: acetonitrile R;
Flow rate 1.5 ml/min.
Detection Spectrophotometer at 230 nm.
Injection 20 µl.
Relative retention With reference to temazepam (retention time = about 16 min): impurity E = about 0.55; impurity F = about 0.67; impurity G = about 0.73; impurity B = about 0.8; impurity D = about 1.2; impurity C = about 1.3; impurity A = about 1.5.
System suitability Reference solution (b):
- — resolution: minimum 1.5 between the peaks due to impurity F and impurity G;
- — peak-to-valley ratio: minimum 1.7, where Hp = height above the baseline of the peak due to impurity G and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to impurity B.
- — correction factors: for the calculation of contents, multiply the peak areas of the following impurities by the corresponding correction factor: impurity F = 3.2; impurity G = 3.1;
- — impurities B, C, D, E, F, G: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
- — any other impurity: for each impurity, not more than 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent);
- — total: not more than 2.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent);
- — disregard limit: 0.25 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C for 4 h.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.250 g in 50 ml of nitroethane R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 ml of 0.1 M perchloric acid is equivalent to 30.07 mg of C16H13ClN2O2.
Protected from light.
Specified impurities A, B, C, D, E, F, G.
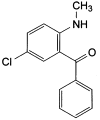
A. [5-chloro-2-(methylamino)phenyl]phenylmethanone,
B. oxazepam,
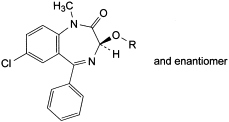
C. R = CO-CH3: (3RS)-7-chloro-1-methyl-2-oxo-5-phenyl-2,3-dihydro-1H-1,4-benzodiazepin-3-yl acetate,
D. R = CH3: (3RS)-7-chloro-3-methoxy-1-methyl-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one,
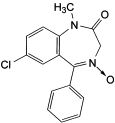
E. 7-chloro-1-methyl-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one 4-oxide,
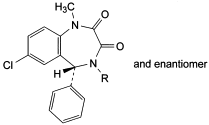
F. R = H: (5RS)-7-chloro-1-methyl-5-phenyl-4,5-dihydro-1H-1,4-benzodiazepine-2,3-dione,
G. R = CH3: (5RS)-7-chloro-1,4-dimethyl-5-phenyl-4,5-dihydro-1H-1,4-benzodiazepine-2,3-dione.
Ph Eur