Sandmeyer Isonitrosoacetanilide Isatin Synthesis
T. Sandmeyer, Helv. Chim. Acta 2, 234 (1919).
Formation of isonitrosoacetodiphenylamidine by condensation of chloral hydrate, hydroxylamine and aniline, cyclization with concentrated sulfuric acid, and quantitative hydrolysis to isatin on dilution:
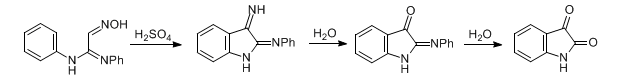
J. Martinet, P. Cousset, Compt. Rend. 172, 1234 (1921); C. Hollins, The Synthesis of Nitrogen Ring Compounds (London, 1924) p 103; C. S. Marvel, G. S. Hiers, Org. Synth. coll. vol. I, 327 (1943); P. L. Julian et al., Heterocycl. Compd. 3, 208 (1952); F. E. Sheibley, J. S. McNulty, J. Org. Chem. 21, 171 (1956); O. Bayer, W. Eckert, Houben-Weyl 7/4, 14 (1968); S. J. Garden et al., Tetrahedron Lett. 38, 1501 (1997). Cf. Sandmeyer Diphenylurea Isatin Synthesis.