Fischer Peptide Synthesis
E. Fischer, Ber. 36, 2982 (1903).
Formation of polypeptides by treatment of an α-chloro or α-bromo acyl chloride with an amino acid ester, hydrolysis to the acid and conversion to a new acid chloride which is again condensed with a second amino acid ester, and so on. The terminal chloride is finally converted to an amino group with ammonia:
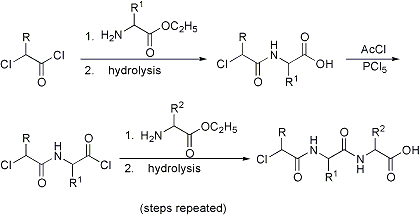
C. L. A. Schmidt, The Chemistry of the Amino Acids and Proteins (Thomas, Springfield, IL, 1944) p 257; B. Rockland in Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) p 232; H. D. Springall, The Structural Chemistry of Proteins (New York, 1954) p 24. Cf. Bergmann-Zervas Carbobenzoxy Method.