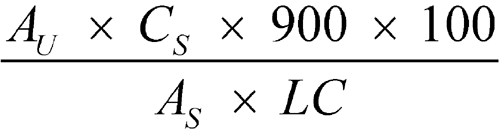Capecitabine Tablets
» Capecitabine Tablets contain not less than 93.0 percent and not more than 105.0 percent of the labeled amount of capecitabine (C15H22FN3O6).
Packaging and storage—
Preserve in tight containers. Store at controlled room temperature.
USP Reference standards  11
11 —
—
USP Capecitabine RS .
.
USP Capecitabine Related Compound A RS .
.
USP Capecitabine Related Compound B RS .
.
USP Capecitabine Related Compound C RS .
.
USP Capecitabine RS
 .
.
USP Capecitabine Related Compound A RS
 .
.
USP Capecitabine Related Compound B RS
 .
.
USP Capecitabine Related Compound C RS
 .
.
Identification—
A:
Infrared Absorption  197K
197K —Bands in the region of 1500–1760 cm
—Bands in the region of 1500–1760 cm 1 obtained for the test sample correspond to the bands obtained for USP Capecitabine RS.
1 obtained for the test sample correspond to the bands obtained for USP Capecitabine RS.
Test specimen—
Grind one Tablet to a fine powder with a mortar and pestle. Mix 1 mg of this sample with 300 mg of potassium bromide.
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Dissolution  711
711 —
—
Medium:
water; 900 mL, degassed.
Apparatus 2:
50 rpm.
Time:
30 minutes.
Standard solution—
For Tablets labeled to contain 150 mg: Transfer 17 mg, accurately weighed, of USP Capecitabine RS to a 100-mL volumetric flask, dissolve in and dilute with Medium to volume, and mix. For Tablets labeled to contain 500 mg: Transfer 28 mg, accurately weighed, of USP Capecitabine RS to a 50-mL volumetric flask, dissolve in and dilute with Medium to volume, and mix.
Test solution—
Pass a portion of the solution under test through a 0.45-µm fiberglass filter.
Procedure—
Determine the amount of capecitabine (C15H22FN3O6) dissolved by employing UV absorption at the wavelength of maximum absorbance at about 304 nm (for Tablets labeled to contain 150 mg) and at about 325 nm (for Tablets labeled to contain 500 mg) on portions of the Test solution, suitably diluted with Medium, if necessary, in comparison with the appropriate Standard solution, using a 1-mm quartz cell. Calculate the amount, in percentage, of C15H22FN3O6 dissolved by the formula:
in which AU and AS are the absorbances obtained from the Test solution and the Standard solution, respectively; CS is the concentration, in mg per mL, of capecitabine (C15H22FN3O6) in the Standard solution; 900 is the volume, in mL, of Medium; 100 is the conversion factor to percentage; and LC is the Tablet label claim, in mg.
Tolerances—
Not less than 80% (Q) of the labeled amount of C15H22FN3O6 is dissolved in 30 minutes.
Uniformity of dosage units  905
905 :
meet the requirements.
:
meet the requirements.
Related compounds—
Diluent, Solution A, Solution B, Peak identification solution, and Chromatographic system—
Proceed as directed in the Assay.
Standard solution—
Use the Standard preparation, prepared as directed in the Assay.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the peak responses. Calculate the percentage of each impurity in the portion of Tablets taken by the formula:
100(1/F)(CS / CU)(ri / rS)
in which F is the relative response factor for each impurity obtained from Table 1; CS is the concentration, in mg per mL, of USP Capecitabine RS in the Standard solution; CU is the concentration, in mg per mL, of capecitabine, based on the label claim, in the Test solution; ri is the peak response for each impurity obtained from the Test solution; and rS is the peak response for capecitabine obtained from the Standard solution. The limits are given in Table 1.
Table 1
| Compound | Relative Retention Time | Relative Response Factor (F) | Limit (%) |
| Capecitabine related compound A | 0.18 | 1.05 | |
| Capecitabine related compound B | 0.19 | 0.81 | |
| Capecitabine | 1.00 | 1.00 | — |
| Capecitabine related compound C | 1.11 | 0.91 | |
| Individual unspecified impurity | — | 1.00 | |
| Total unspecified impurities | — | — | |
| Total impurities | — | — |
Assay—
Diluent—
Prepare a mixture of water, methanol, and acetonitrile (60:35:5).
Diluted acetic acid—
Prepare a 0.1% (v/v) mixture of acetic acid in water.
Solution A—
Prepare a mixture of Diluted acetic acid, methanol, and acetonitrile (60:35:5).
Solution B—
Prepare a mixture of methanol, Diluted acetic acid, and acetonitrile (80:15:5).
Standard preparation—
Dissolve an accurately weighed quantity of USP Capecitabine RS in Diluent, and sonicate if necessary, to obtain a solution having a known concentration of about 0.6 mg per mL.
Peak identification solution—
Dissolve suitable quantities of USP Capecitabine RS, USP Capecitabine Related Compound A RS, USP Capecitabine Related Compound B RS, and USP Capecitabine Related Compound C RS in Diluent, and sonicate if necessary, to obtain a solution having known concentrations of about 0.06 mg of each per mL. Quantitatively dilute this solution with Diluent to obtain a solution having known concentrations of about 0.6 µg of each per mL.
Assay preparation—
Grind not fewer than 20 Tablets to a fine powder. Dissolve an accurately weighed quantity of powdered Tablets in Diluent, dilute quantitatively with Diluent, and sonicate if necessary, to obtain a solution having a known concentration of about 0.6 mg per mL of capecitabine, based on the label claim. Pass through a PVDF 0.45-µm membrane filter, and use the filtrate.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 250-nm detector, a refrigerated autosampler maintained at 5
)—
The liquid chromatograph is equipped with a 250-nm detector, a refrigerated autosampler maintained at 5 , and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 40
, and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 40 . The chromatograph is programmed as follows.
. The chromatograph is programmed as follows.
Chromatograph the Diluent and the Peak identification solution, and record the peak responses as directed for Procedure: the resolution, R, between capecitabine related compound A and capecitabine related compound B is not less than 1.0. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the tailing factor of the capecitabine peak is not greater than 1.5; and the relative standard deviation for replicate injections of the capecitabine peak is not more than 2.0%. [note—For the purpose of peak identification the approximate relative retention times are given in Table 1. The relative retention times are measured with respect to capecitabine.]
| Time (minutes) |
Solution A (%) | Solution B (%) | Elution |
| 0–5 | 100 | 0 | isocratic |
| 5–20 | 100®49 | 0®51 | linear gradient |
| 20–30 | 49 | 51 | isocratic |
| 30–31 | 49®100 | 51®0 | linear gradient |
| 31–40 | 100 | 0 | equilibration |
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the capecitabine peaks. Calculate the quantity, in percentage of label claim, of C15H22FN3O6 in the portion of Tablets taken by the formula:
100 (CS / CU)(rU / rS)
in which CS is the concentration, in mg per mL, of USP Capecitabine RS in the Standard preparation; CU is the concentration, in mg per mL, of capecitabine, based on the label claim, in the Assay preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Feiwen Mao, M.S.
Scientist 1-301-816-8320 |
(MDOOD05) Monograph Development-Ophthalmics Oncologics and Dermatologicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
|
| Margareth R.C. Marques, Ph.D.
Senior Scientist 1-301-816-8106 |
(BPC05) Biopharmaceutics05 |
USP32–NF27 Page 1775
Pharmacopeial Forum: Volume No. 32(4) Page 1054
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.