Nenitzescu Reductive Acylation
C. D. Nenitzescu, E. Cioranescu, Ber. 69, 1820 (1936).
Hydrogenative acylation of cycloolefins with acid chlorides in the presence of aluminum chloride; with five- and six-membered rings no change in ring size occurs but with seven-membered rings rearrangement takes place with formation of a cyclohexane derivative:
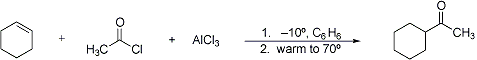
C. Nenitzescu, C. N. Ionescu, Ann. 491, 189 (1931); C. D. Nenitzescu, J. P. Cantuniari, ibid. 510, 269 (1934); C. D. Nenitzescu, I. Chicos, Ber. 68, 1584 (1935); C. A. Thomas, Anhydrous Aluminum Chloride in Organic Chemistry (New York, 1941) p 759; S. L. Friess, R. Pinson, J. Am. Chem. Soc. 73, 3512 (1951); Olah, Friedel-Crafts and Related Reactions vol. III, Part 2 (New York, 1964) p 1069. Cf. Darzens-Nenitzescu Synthesis of Ketones.